Breakthrough in the surgical treatment of glioblastoma
- Glioblastoma continues to pose a major clinical challenge because of its aggressive nature, limited therapeutic options, and poor prognosis.
- Specialists in Łódź have introduced an innovative surgical approach using 18F-FET PET imaging, allowing more accurate identification of active tumor tissue during resection.
- Alongside advances in surgery, ongoing research into monoclonal antibodies, checkpoint inhibitors, and ADCs highlights the growing role of biological therapies in the search for more effective treatment.
Pioneering surgery at a hospital in Łódź
Glioblastoma is one of the most aggressive brain tumors. Specialists from a The Nicolaus Copernicus Provincial Multispecialty Center for Oncology and Traumatology in Łódź have performed the first surgery in Poland to remove the tumor using PET imaging with 18-fluorotyrosine (18F-FET).
During the procedure, the patient receives the 18F-FET. This substance becomes involved in the tumor’s metabolic processes and participates in protein synthesis. As a result, physicians can more precisely identify areas where tumor activity is higher than in healthy brain tissue.
Until now, no glioblastoma removal surgery in Poland had been planned using PET imaging. The first patient who underwent surgery using the new method on March 6, is feeling very well and is already ready for the next stage of his fight against the disease.
Doctors at the hospital in Łódź are currently recruiting patients with newly diagnosed glioblastoma who would like to participate in an innovative treatment program. Interested individuals can apply directly to the Department of Neurosurgery and Tumors of the Nervous System via mail or phone. The all procedures was performed as part of a scientific study funded by the Medical Research Agency.
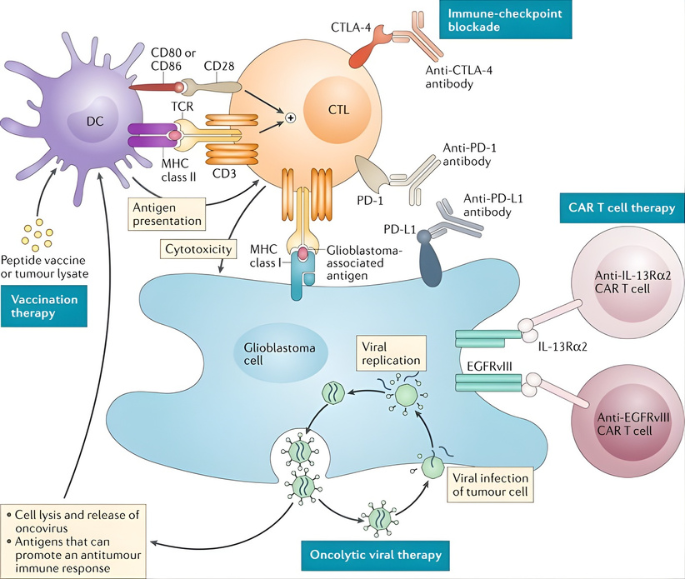
Immunotherapy used to cure glioblastoma
Current approaches to the treatment of glioblastoma include surgery, chemotherapy, and radiotherapy.
Surgery is not always possible, and systemic treatment is associated with well-known risks. The only approved biological therapy is bevacizumab (Avastin). This monoclonal antibody can help reduce tumor size or relieve swelling and symptoms in some patients, particularly in cases where the cancer has recurred after treatment. New targeted therapies are currently being investigated in clinical trials, including novel drugs, new therapeutic combinations, and innovative treatment approaches. These studies give patients the opportunity to access the latest solutions that may prove to be effective.
Glioblastoma remains a difficult target for biological therapy. The core problem is not lack of targetable biology, but a mismatch between mechanisms of action and hostile clinical reality. At present, the antibody class with the clearest clinical utility in glioblastoma is anti-VEGF therapy, checkpoint inhibitors and EGFR-directed antibodies. ADCs have shown mechanistic promise without producing an approved standard.
Glioblastoma is the most common and lethal type of tumor of the central nervous system, with an average survival of 15 months after first diagnosis. According to the National Brain Tumor Society, the five-year survival rate for patients is only 6.8%. Let us join forces to deliver a solution to the market that will help patients overcome this cancer.
Prepared by:

Marketing Specialist
Sources and further reading
- Kopernik. Pierwsza w Polsce operacja. 2026.
- Sarfraz Z, Maharaj A, Venur VA, Lathia JD, Odia Y, Ahluwalia MS. Immunotherapy in Glioblastoma: An Overview of Current Status. Clin Pharmacol. 2025; 17: 185-209.
- Liu Y, Zhou F, Ali H, Lathia JD, Chen P. Immunotherapy for glioblastoma: current state, challenges, and future perspectives. Cell Mol Immunol. 2024; 21(12): 1354-1375.
- Yu Z, Zhao G, Zhang Z, Li Y, Chen Y, Wang N, Zhao Z, Xie G. Efficacy and safety of bevacizumab for the treatment of glioblastoma. Exp Ther Med. 2016; 11(2): 371-380.
- Parakh S, Nicolazzo J, Scott AM, Gan HK. Antibody Drug Conjugates in Glioblastoma – Is There a Future for Them? Front Oncol. 2021; 11: 718590.
- Lim M, Xia Y, Bettegowda C, Weller M. Current state of immunotherapy for glioblastoma. Nat Rev Clin Oncol. 2018; 15(7): 422-442.
- Kesumayadi I, Kambe A, Kanda H, Hosoya T, Sakamoto M, Kurosaki M. Bevacizumab enhances overall survival in newly diagnosed glioblastoma patients with high COX-2 expression. Sci Rep. 2026; 16(1): 4974.
- Zoi V, Galani V, Sioka C, Alexiou GA, Kyritsis AP. Immunotherapy for Glioblastoma: A Focus on PD-1/PD-L1 Inhibitors. Cancers (Basel). 2025; 17(23): 3777.
- Manzarbeitia-Arroba B, Hodolic M, Pichler R, Osipova O, Soriano-Castrejón ÁM, García-Vicente AM. 18F-Fluoroethyl-L Tyrosine Positron Emission Tomography Radiomics in the Differentiation of Treatment-Related Changes from Disease Progression in Patients with Glioblastoma. Cancers (Basel). 2023; 16(1): 195.