Science News
Discover the news and inspiring materials from the world of biotechnology.

Mabion Biologics at mBank Healthcare Days 2026
Mabion C-suite participated in a meeting with investors and stakeholders during mBank Healthcare Days 2026 in Warsaw. Mabion is engaged in advanced licensing, co-development, and consortium discussions, the results of which will be announced in the H1 2026.
Learn more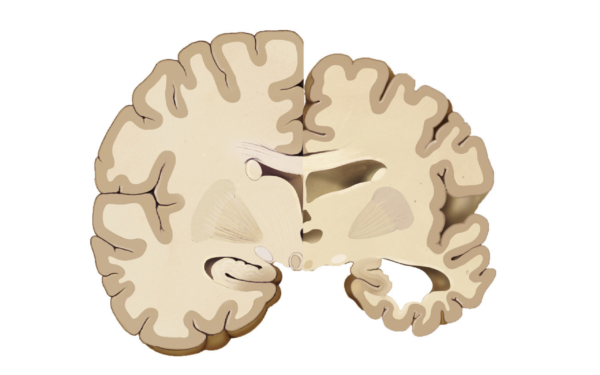
New Era in Alzheimer’s Disease Antibody Treatment
New antibody treatments are redefining Alzheimer’s care. Lecanemab and donanemab target amyloid plaques and show measurable clinical benefits. Explore how safe and effective these therapies really are.
Learn more
The PLN 5.6 Billion Bill for COVID-19 Vaccines
A Brussels court has ordered Poland to pay over 5.6 billion PLN to Pfizer. Behind the ruling lies a procurement catastrophe and a hard lesson in what happens when a state bets against its own industrial base.
Learn more
Executing Mabion’s CDMO Vision: Insights from CEO Gregor Kawaletz
Gregor Kawaletz, CEO of Mabion Biologics CDMO, discusses how Mabion is executing its transformation into a specialized CDMO partner for biologic drug substance development and manufacturing.A key message of the conversation is Mabion’s focus on building long-term value through deeper collaboration with biotechnology and pharmaceutical partners. As the CDMO market evolves, the company is strengthening its position not only as a service provider, but also as a strategic partner engaged in technology development, process advancement, and co-development projects.
Learn more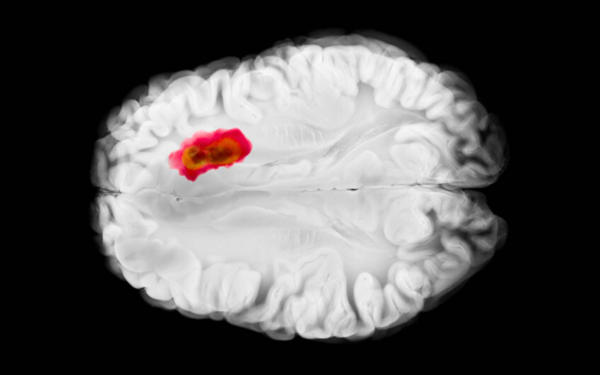
Breakthrough in the surgical treatment of glioblastoma
Doctors at a hospital in Łódź have performed Poland’s first surgery to remove a glioblastoma using PET imaging with 18-fluorotyrosine, a technique currently used in only a few neurosurgical centers worldwide. Additional patients are now being sought who could undergo this innovative therapy.
Learn more
Establishing Business Connections with Polish Entrepreneurs
Industry platforms provide a valuable environment where innovators, CDMOs, and technology providers can build relationships that ultimately translate into impactful collaborations and new biopharma projects. Business connections will allow you to find non-obvious synergies. Take a closer look to business networking events in Łódź.
Learn more
Gregor Kawaletz featured on Making It in Manufacturing podcast
Key topics include Mabion’s growth priorities, confidence-building across the pipeline, and an evolving business model designed to support long-term value creation.
Learn more
How Mabion Builds Space for Women in Biopharma
Through the stories of Klaudia Lechowska and Natalia Wojnowska, this feature explores how leadership trust, internal opportunity, and cross-functional growth enable women to thrive in both laboratory and business roles. At Mabion Biologics CDMO, supporting women in science goes far beyond celebration days. It is embedded in career mobility, mentorship, and structured development. Discover how an environment built on support, visibility, and scientific ambition helps transform potential into lasting impact.
Learn more
Mentoring program supports industry–academia collaboration
The mentoring program “Siła Wspólnoty” creates space for long-term industry–academia collaboration. The second edition of the program featured 34 mentors, including a dozen who also participated in the first edition of the initiative, and 39 mentees.
Learn more