New Era in Alzheimer’s Disease Antibody Treatment
- For decades, the search for an effective Alzheimer’s disease antibody treatment was limited to managing symptoms, with traditional alternatives offering only temporary boosts to memory without addressing the underlying brain destruction.
- The landscape has fundamentally shifted with the arrival of lecanemab (Leqembi) and donanemab (Kisunla). These monoclonal antibodies represent the first disease-modifying therapies to receive traditional FDA approval.
- Recent research suggests new strategies for combining anti-amyloid monoclonal antibodies and small molecules for the treatment of Alzheimer’s disease.
How does Anti-Amyloid Antibody Treatment Work?
The primary goal of anti-amyloid antibody therapy is to remove beta-amyloid protein deposits from the brain. There are currently two monoclonal antibodies (lecanemab and donanemab) on the market that have passed the approval process by the regulatory agencies FDA and EMA (although Lecanemab was initially rejected, as we reported in this article).
The fundamental theory behind these treatments is the amyloid cascade hypothesis, which proposes that the accumulation of amyloid beta is the central event that leads to the neurofibrillary tangles and cell death characteristic of Alzheimer’s. By clearing these proteins, the drugs aim to slow the clinical progression of the disease, allowing patients in the early stages to maintain their quality of life for a longer period.
Lecanemab (Leqembi) therapy primarily targets soluble amyloid-beta protofibrils, which are toxic precursors that eventually clump together to form hard plaques. A Fc region of the antibody acts as a biological switch. When lecanemab binds to the amyloid, this fragment signals the brain’s own immune cells, called microglia, to latch on and clear the toxic deposits through cellular processes like phagocytosis and lysosomal activity.
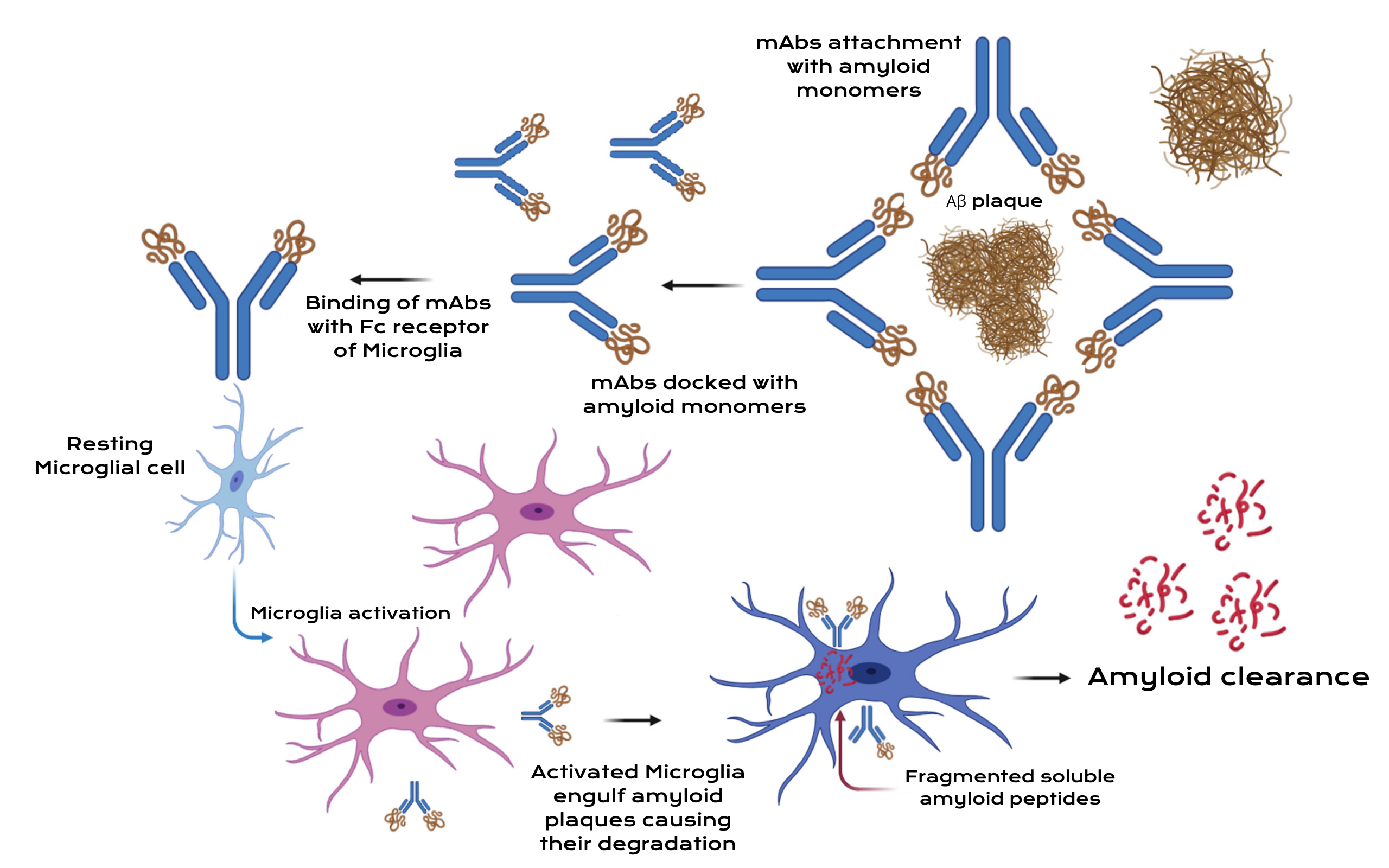
Donanemab (Kisunla) is a “plaque-specific” antibody that targets an insoluble, modified, N-terminal truncated form of amyloid that is only found in established brain plaques. By binding specifically to these mature deposits, it facilitates their removal through microglial-mediated phagocytosis, essentially scrubbing existing plaques from the brain tissue.
Effectiveness and Safety Profile of Alzheimer’s Disease Antibody Treatment
The effectiveness and Safety Profile of these therapies has been validated in large-scale Phase 3 clinical trials, showing a significant slowing of cognitive and functional decline in patients at the early stages of the disease.
- Clarity AD (Lecanemab): In a trial of 1,795 participants, lecanemab slowed the speed of memory and thinking decline by 27% over 18 months compared to placebo. Perhaps even more importantly for patients and families, the drug reduced the loss of quality of life by up to 56%.
- TRAILBLAZER-ALZ 2 (Donanemab): This trial demonstrated that donanemab slowed clinical progression by 35.1% in patients with low-to-medium levels of tau and 22.3% in a combined population. For many, this resulted in a delay in symptom progression of roughly 4.5 to 7.5 months over an 18-month period. In the donanemab trials, 80.1% of participants in the low/medium tau population achieved amyloid clearance by 76 weeks, meaning their plaque levels dropped to a point where they were effectively “amyloid negative”.
Safety is managed through precision monitoring for Amyloid-Related Imaging Abnormalities (ARIA): small areas of brain swelling (ARIA-E) or microbleeds (ARIA-H). In clinical trials, ARIA occurred in 24.0% of donanemab patients and 12.6% of lecanemab patients. Crucially, the majority of these cases (roughly 75%) were asymptomatic and only detectable via MRI.
New Approach to Combination Therapy using Antibodies and Small Molecules
Recent study that pairing anti-amyloid antibodies with compounds derived from resveratrol and curcumin significantly enhances amyloid clearance. In laboratory models, antibody monotherapy reduced amyloid aggregation by roughly 50-71%, whereas combination therapy achieved up to 89-97% inhibition, indicating a strong synergistic effect.
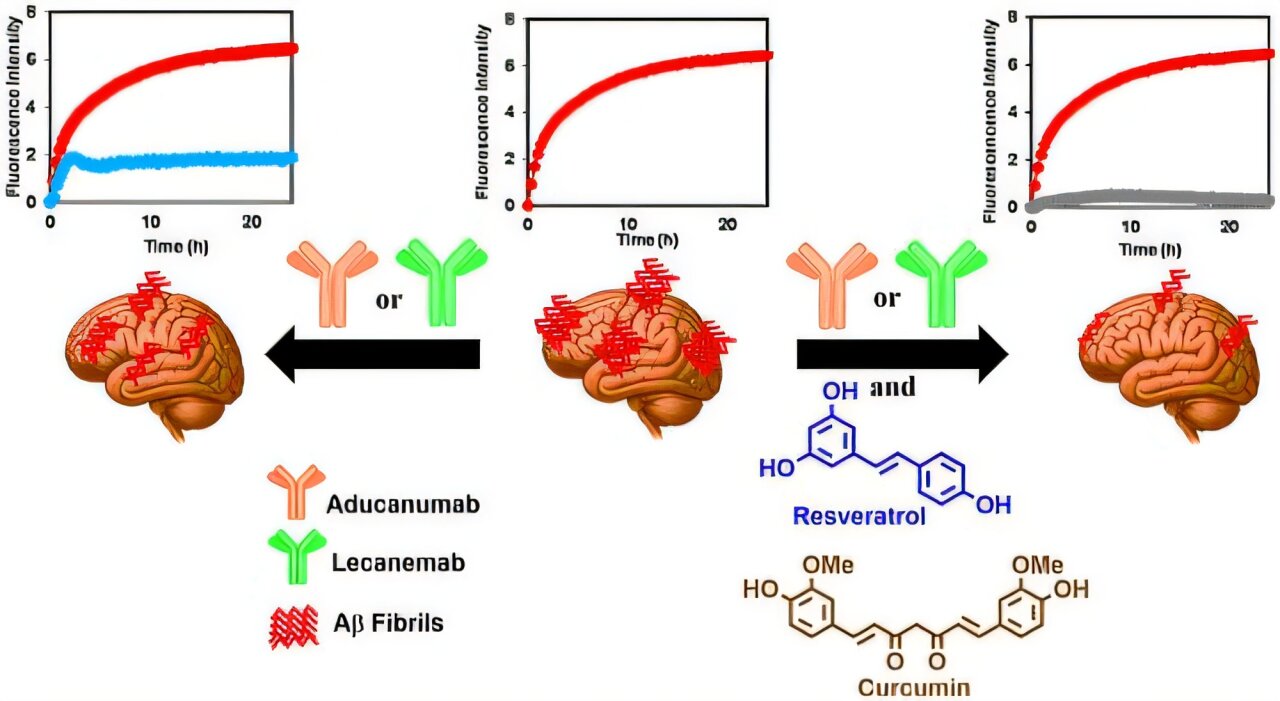
The combination therapy may also substantially improve safety. By enhancing therapeutic potency, researchers propose that lower doses of monoclonal antibodies could be used, potentially reducing the risk of adverse events like ARIA (brain swelling or bleeding). Early cellular studies also showed that these combinations were non-toxic to neurons and reduced amyloid-induced cell damage more effectively than antibodies alone, supporting both efficacy and tolerability.
These findings suggest that small molecules and antibodies act on complementary binding sites, enabling more efficient disruption of toxic protein aggregation.

If you are interested in our services regarding the monoclonal antibodies manufacturing, please don’t hesitate to contact us. Mabion’s specialist offer individualized approach tailored to the biologic and clinical characteristics of the developed drug. Our experience with development and manufacturing of biologics gives us an overwhelming advantage over other CDMOs as we perfectly understand the client’s perspective.
Click here to obtain more information on our services.
Prepared by:

Marketing Specialist
Sources
- Le Boeuf W, Hefny AA, Karuturi RC, Rao PN. Combination of Resveratrol and Curcumin with Anti-Amyloid Monoclonal Antibodies Aducanumab and Lecanemab Leads to Greater Inhibition of Amyloid-Beta Aggregation. ACS Chem. Neurosci. 2026; 17(5): 987-997.
- Cummings J. Anti-Amyloid Monoclonal Antibodies are Transformative Treatments that Redefine Alzheimer’s Disease Therapeutics. Drugs. 2023; 83(7): 569-576.
- Alkhalifa AE, Al Mokhlf A, Ali H, Al-Ghraiybah NF, Syropoulou V. Anti-Amyloid Monoclonal Antibodies for Alzheimer’s Disease: Evidence, ARIA Risk, and Precision Patient Selection. J Pers Med. 2025; 15(9): 437.
- Abduljawad AA, Alkinani KB, Zaakan A, AlGhamdi AS, Hamdoon AAE, Alshanbari BH, Alshehri AA, Alluhaybi BB, Alqashi SOI, Abduljawad RA. Targeting Amyloid-β Proteins as Potential Alzheimer’s Disease Therapeutics: Anti-Amyloid Drug Discovery, Emerging Therapeutics, Clinical Trials and Implications for Public Health. Pharmaceuticals (Basel). 2025; 18(11): 1731.
- Zimmer JA, Sims JR, Evans CD, Nery ESM, Wang H, Wessels AM, Tronchin G, Sato S, Raket LL, Andersen SW, Sapin C, Paget MA, Gueorguieva I, Ardayfio P, Khanna R, Brooks DA, Matthews BR, Mintun MA; Alzheimer’s Disease Neuroimaging Initiative. Donanemab in early symptomatic Alzheimer’s disease: results from the TRAILBLAZER-ALZ 2 long-term extension. J Prev Alzheimers Dis. 2026; 13(2): 100446.
- Sims JR, Zimmer JA, Evans CD, Lu M, Ardayfio P, Sparks J, Wessels AM, Shcherbinin S, Wang H, Monkul Nery ES, Collins EC, Solomon P, Salloway S, Apostolova LG, Hansson O, Ritchie C, Brooks DA, Mintun M, Skovronsky DM; TRAILBLAZER-ALZ 2 Investigators. Donanemab in Early Symptomatic Alzheimer Disease: The TRAILBLAZER-ALZ 2 Randomized Clinical Trial. JAMA. 2023; 330(6): 512-527.
- Taylor E. New Alzheimer’s drug, donanemab – what is it and how does it work? Alzheimer’s Research UK. 2024.
- van Dyck CH, Swanson CJ, Aisen P, Bateman RJ, Chen C, Gee M, Kanekiyo M, Li D, Reyderman L, Cohen S, Froelich L, Katayama S, Sabbagh M, Vellas B, Watson D, Dhadda S, Irizarry M, Kramer LD, Iwatsubo T. Lecanemab in Early Alzheimer’s Disease. N Engl J Med. 2023; 388(1): 9-21.
- Yang HM. Recent Advances in Antibody Therapy for Alzheimer’s Disease: Focus on Bispecific Antibodies. Int J Mol Sci. 2025; 26(13): 6271.
- Puranik N, Kumari M, Tiwari S, Dhakal T, Song M. Resveratrol as a Therapeutic Agent in Alzheimer’s Disease: Evidence from Clinical Studies. Nutrients. 2025; 17(15): 2557.