
Biologics

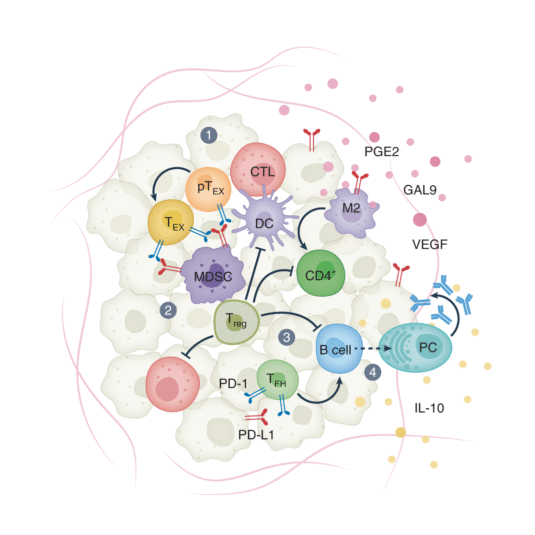
Melanoma Immunotherapy Enhance by Immunity Activation

Single-Dose Monoclonal Antibody Therapy Offers Durable Protection Against Malaria

Biopharma Drug Development Gaps Strategy

Winning Trust Through Mabion Biologics CDMO Excellence – exclusive interview with Marty Henehan

Mabion is a Champion of the 2025 International CDMO Leadership Award in Biologics

Mabion’s Fill & Finish services in Contract Pharma Magazine

Mabion is a Finalist in the CDMO Leadership Awards 2025 in the Biologics category
