Science News
Discover the news and inspiring materials from the world of biotechnology.

Executing Mabion’s CDMO Vision: Insights from CEO Gregor Kawaletz
Gregor Kawaletz, CEO of Mabion Biologics CDMO, discusses how Mabion is executing its transformation into a specialized CDMO partner for biologic drug substance development and manufacturing.A key message of the conversation is Mabion’s focus on building long-term value through deeper collaboration with biotechnology and pharmaceutical partners. As the CDMO market evolves, the company is strengthening its position not only as a service provider, but also as a strategic partner engaged in technology development, process advancement, and co-development projects.
Learn more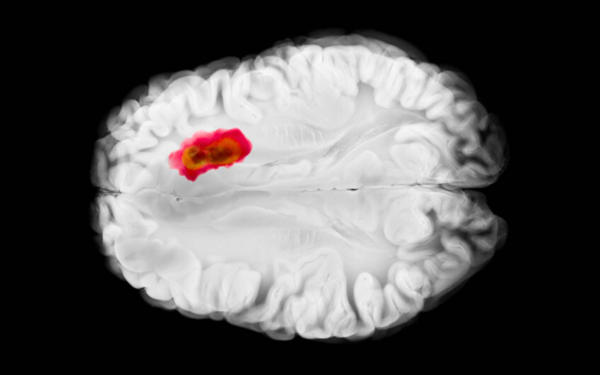
Breakthrough in the surgical treatment of glioblastoma
Doctors at a hospital in Łódź have performed Poland’s first surgery to remove a glioblastoma using PET imaging with 18-fluorotyrosine, a technique currently used in only a few neurosurgical centers worldwide. Additional patients are now being sought who could undergo this innovative therapy.
Learn more
Establishing Business Connections with Polish Entrepreneurs
Industry platforms provide a valuable environment where innovators, CDMOs, and technology providers can build relationships that ultimately translate into impactful collaborations and new biopharma projects. Business connections will allow you to find non-obvious synergies. Take a closer look to business networking events in Łódź.
Learn more
Gregor Kawaletz featured on Making It in Manufacturing podcast
Key topics include Mabion’s growth priorities, confidence-building across the pipeline, and an evolving business model designed to support long-term value creation.
Learn more
How Mabion Builds Space for Women in Biopharma
Through the stories of Klaudia Lechowska and Natalia Wojnowska, this feature explores how leadership trust, internal opportunity, and cross-functional growth enable women to thrive in both laboratory and business roles. At Mabion Biologics CDMO, supporting women in science goes far beyond celebration days. It is embedded in career mobility, mentorship, and structured development. Discover how an environment built on support, visibility, and scientific ambition helps transform potential into lasting impact.
Learn more
Mentoring program supports industry–academia collaboration
The mentoring program “Siła Wspólnoty” creates space for long-term industry–academia collaboration. The second edition of the program featured 34 mentors, including a dozen who also participated in the first edition of the initiative, and 39 mentees.
Learn more
Mabion’s Article about RFP process named one of the Top 10 Industry Insights of 2025 by Outsourced Pharma
As biologics continue to demonstrate their versatility across the pharma landscape, biotechs are looking for capable CDMOs that can bring these therapies to market to provide innovative treatment to patients with chronic diseases. Amid the vast discussion around how best to assess a CDMO’s offerings and determine whether they are equipped to fulfill a drug program’s needs, the importance of optimizing the request for proposal (RFP) is often omitted. Generally, an RFP describes the immediate needs of the proposed project, the services needed from the CDMO, projected timeline, and budget, all of which is used by a CDMO to generate a cost proposal for services to be rendered. Depending on the needs and knowledge of the sponsor, it might also detail specific technologies and key data points to be procured.
Learn more
Declaration of the objectives and scope of the European Biotech Act
The European Commission has announced the European Biotech Act, a major policy initiative aimed at strengthening the EU’s competitiveness in health biotechnology and biopharmaceutical innovation. The move responds to a growing gap between Europe’s strong scientific base and its weaker industrial and financial performance in biotech. Health biotechnology employs more than 900,000 people in the EU, around 75% of them in the health sector, and contributes close to €40 billion annually to the European economy. Despite this, the EU attracts only about 7% of global venture capital investment in health biotech, while its share of global commercial clinical trials has declined from 22% to 12% over the past decade. The Biotech Act is designed to address these challenges by reducing regulatory complexity, accelerating development timelines, and improving access to investment capital for biotech companies operating in Europe.
Learn more
AI Comes of Age in Biologic Drug Discovery
AI drug discovery is rapidly reshaping how large-molecule therapeutics are conceived, moving the industry from empirical screening toward intentional, computational design. Recent advances show that artificial intelligence can now propose entirely new antibody structures with levels of precision and functionality approaching those required for real medicines. These technologies increasingly account not only for target binding but also for properties critical to development, such as stability, specificity, and manufacturability. As a result, biological targets once considered difficult or impractical for large-molecule drugs are coming back into strategic focus. While clinical adoption will still require rigorous validation and safety testing, the trajectory suggests a fundamental shift in how biopharmaceutical innovation will unfold.
Learn more