Integrated Modeling of Upstream and Downstream Bioprocessing to Improve Biologics Production Efficiency
- Biologics production efficiency can be improved when upstream and downstream bioprocessing are planned and optimized together as a single platform.
- Modeling showed how changes in cell culture affect product quality and purification, allowing better planning of the process.
- Mabion’s platform improves process reliability by integrating data-driven modeling.
Introduction
Biopharma manufacturing has evolved significantly, driven by the increasing demand for monoclonal antibodies and other complex biologics. Despite major advances in cell line engineering, media development, and purification technologies, many production processes are still optimized in a fragmented way, with upstream and downstream operations treated as separate entities. This separation often leads to suboptimal process performance, higher costs, and limited process robustness.
Integrated bioprocess modeling allows process scientists to understand how changes in cell culture conditions influence product quality attributes and purification performance. By combining experimental data from upstream process development with downstream purification models, it became possible to anticipate process bottlenecks and optimize both productivity and product quality simultaneously.
Upstream Processing
Upstream bioprocessing for biologics typically relies on mammalian cell lines, most commonly CHO cells. Extensive optimization was performed to evaluate the effects of feeding strategies and metabolic control on productivity. Monitoring metabolites provided insights into cellular metabolism and process stability. Process modeling helped identify correlations between nutrient availability and antibody productivity.
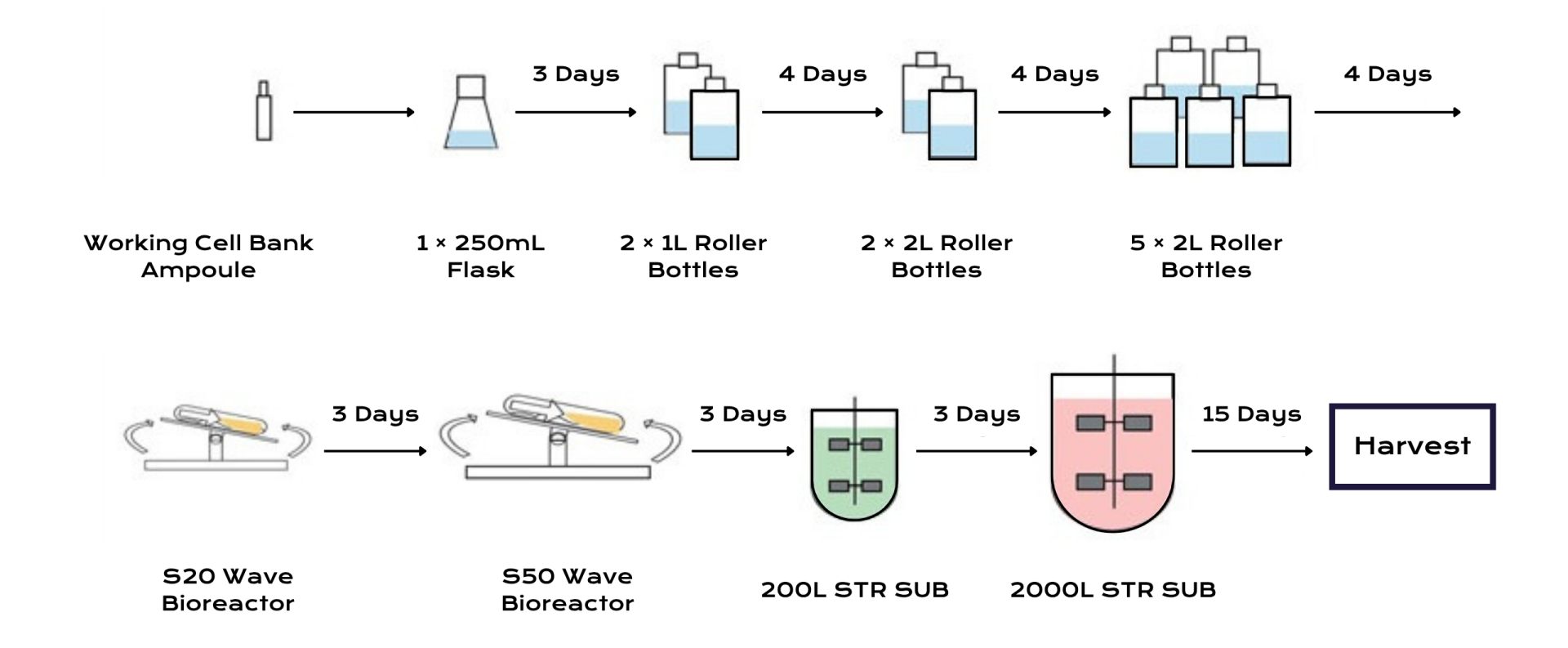
These models were then used to design feeding strategies that reduced metabolic stress and improved overall yield.
Downstream Processing
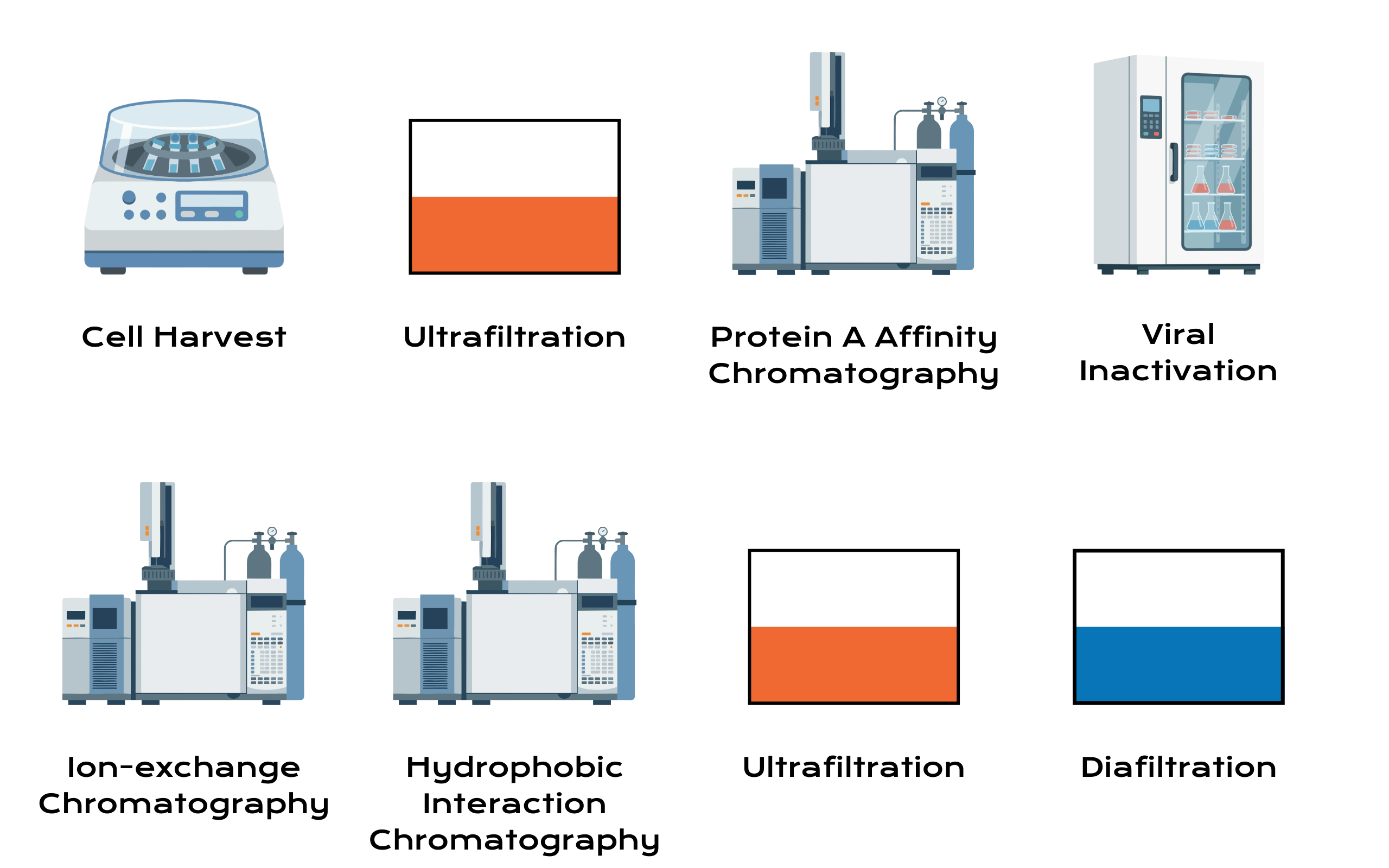
Downstream bioprocessing is responsible for isolating the target protein from complex cell culture harvests while ensuring product purity, safety, and quality. Our purification process begins with Protein A affinity chromatography, which captures antibodies with high specificity and efficiency. This step is followed by additional chromatography polishing operations, designed to remove impurities.
Biologics Production Efficiency Platform
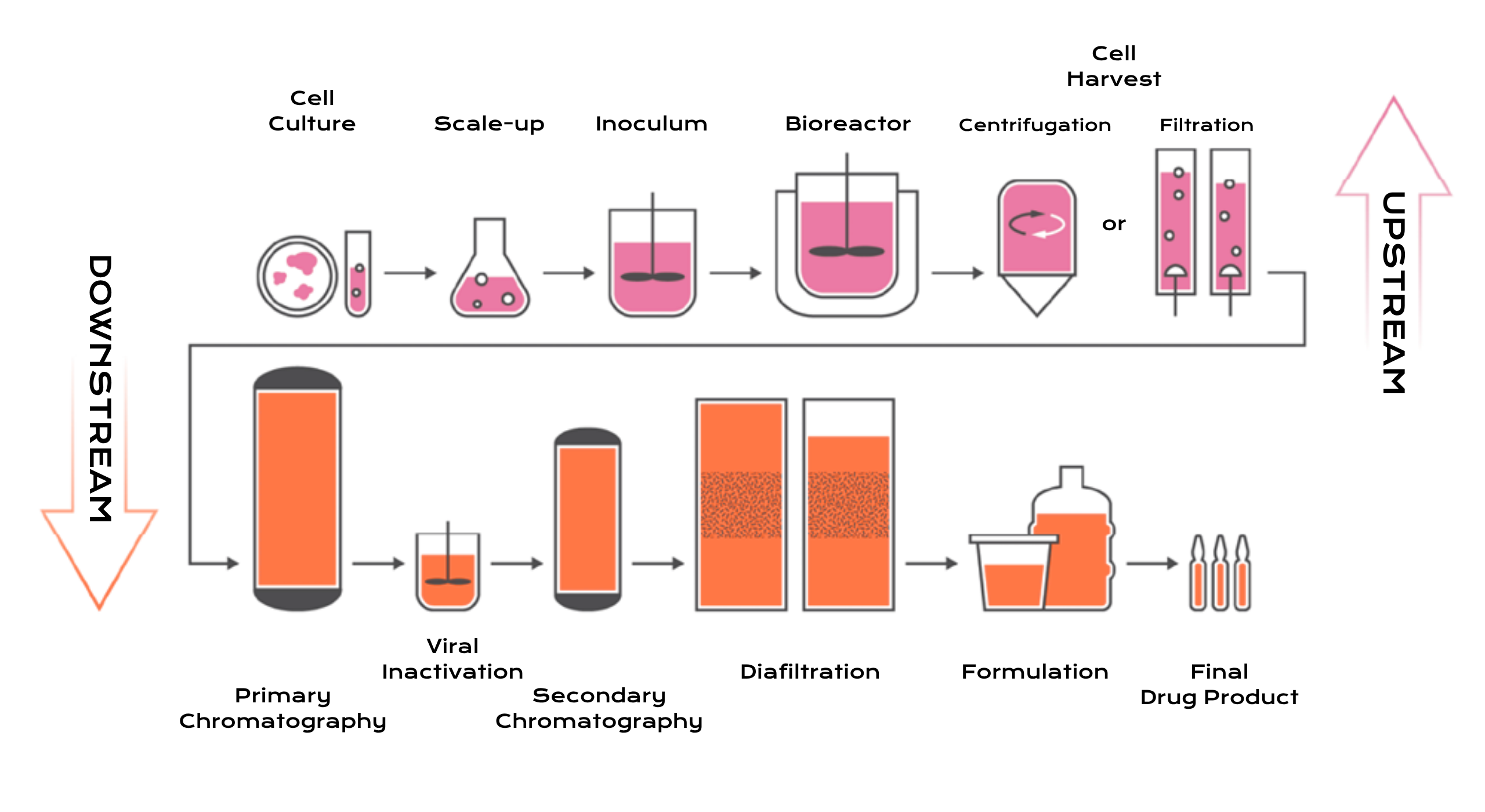
A platform approach to biologics manufacturing involves developing standardized process frameworks that can be applied across multiple monoclonal antibody products. Such platforms accelerate development timelines, reduce technical risk, and enable more predictable drug substance manufacturing performance. They allow rapid onboarding of new client molecules while maintaining consistent quality and regulatory compliance.
Summary
The integration of upstream and downstream process modeling represents an important step toward more data-driven and predictive biopharma manufacturing. Using CHO as an expression model system, upstream process modeling allowed the identification of feeding strategies that enhanced productivity while minimizing metabolic stress. These improvements directly influenced downstream processing by modifying impurity loads and product quality attributes. The downstream platform built around affinity and polishing chromatography provides a robust purification strategy capable of handling varying upstream conditions. When combined with upstream data and modeling tools, this platform supports faster process development and more predictable scale-up.
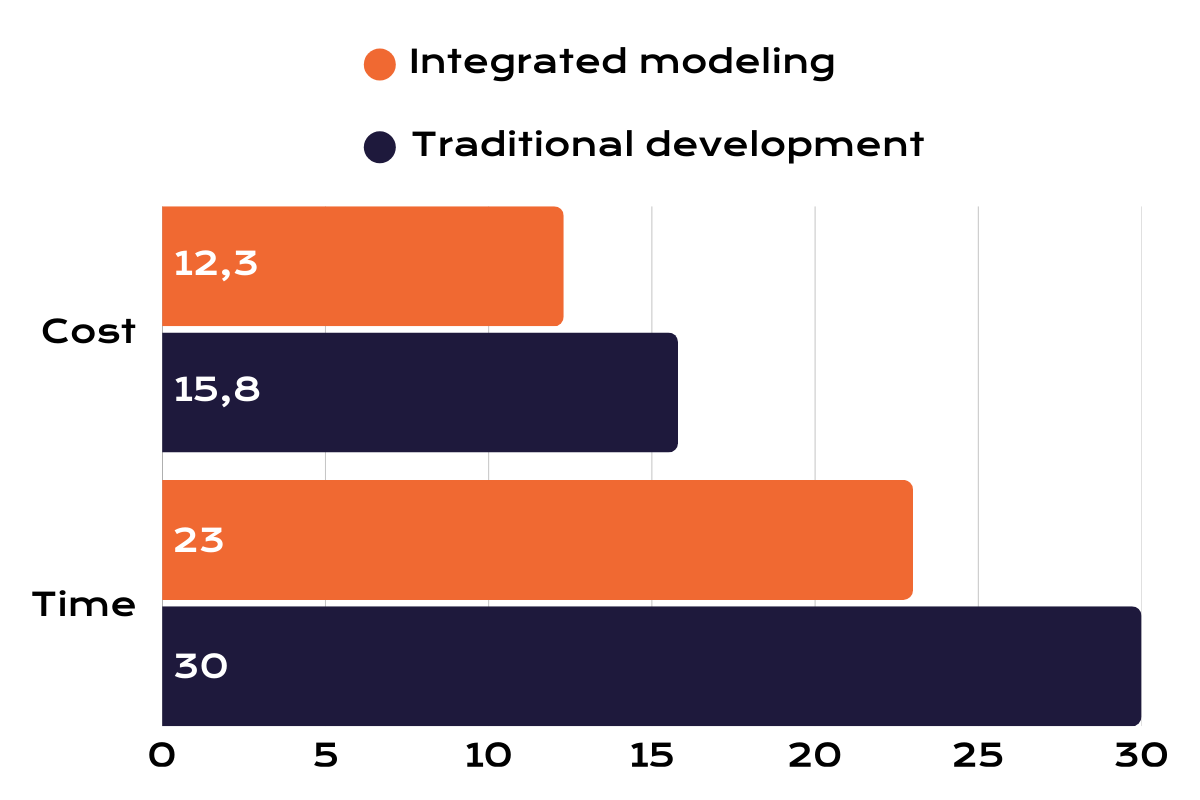
As illustrated in the development time and cost comparison, integrated modeling can significantly reduce process development timelines and experimental workload while improving overall biologics production efficiency.
FAQ
Prepared by:

Marketing Specialist

Business Development Specialist
References
- Balcerek J, Bednarek M, Sobieściak TD, Pietrucha T, Jaros S. Toward Shortened the Time-to-Market for Biopharmaceutical Proteins: Improved Fab Protein Expression Stability Using the Cre/lox System in a Multi-Use Clonal Cell Line. J Pharm Sci. 2021; 110(2): 946-951.
- Nosek M, Grzyb O, Tuszyner A. Metabolite and nutrient analysis as crucial components for optimal CHO cell culture upstream process. Mabion Science Hub. 2023.
- Morawska A, Miłek P, Urbaniak M. Ion Exchange Chromatography in Monoclonal Antibodies Purification. Mabion Science Hub. 2024.
- Guajardo N, Schrebler RA. Upstream and Downstream Bioprocessing in Enzyme Technology. Pharmaceutics. 2023; 16(1): 38.
- Lonkwic K, Moskal A, Knurek J. CHO Cell Lines for Recombinant Protein Production. Mabion Science Hub. 2025.

If you are interested in our services regarding the biologics production efficiency, please don’t hesitate to contact us. Mabion’s specialist offer individualized approach tailored to the biologic and clinical characteristics of the developed drug. Our experience with end-to-end development and manufacturing of biologics gives us an overwhelming advantage over other CDMOs as we perfectly understand the client’s perspective. Click here to obtain more information on our services.